|
As more protons are added to the nucleus, they have a stronger attraction to the electrons. Atomic radius, however, will decrease when moving left to right. English: Showing how ionization energy increases as you go up or to the right on the Periodic Table. Moving left to right on the periodic table causes an increase in atomic number (number of protons) as well as electron affinity and electronegativity. Therefore, the lowest ionisation energy will be Francium, and the highest ionisation energy will be Helium. Add a one-line explanation of what this file represents. Ionisation energy will increase from left to right, and it will rise from the bottom to the top on the periodic table. Metals have low ionisation energy, whereas nonmetals have high ionisation energy. However, turning the element into gas is necessary before moving onto the electron. With the help of Ionisation, one can ionise different elements in the periodic table and turn them into cations. When fluorine gains an electron, it will have a negative charge. Cation is just another word for positive ions. Below is a diagram showing the directions atomic size increase over the periodic table. The IE increase from bottom to top and left to right in the periodic table. And so, when it’s a positive ion where the number of protons is more than electrons, it is called Cations. The equation for the second ionization energy is: Na +-> Na 2+ + e-Ionization energy values are typically very high and follow trends throughout the periodic table. Still, even if one of the electrons is taken away, then Hydrogen would have a positive charge, and essentially, it would just be, in its most common isotope, it would just be a proton by itself. If we spectate on the periodic table and travel from left to right across the elements, the ionization energy will rise due to a declining atomic radius. So, for example, Hydrogen in its neutral state has one proton and one electron. In the periodic table, the ionization energy of an electron upsurge with the atomic number of the atom and drops for higher energy orbitals. The negative ions are more significant in the number of electrons than protons. A molecule’s just a cluster of atoms bonded together. The charge is given from protons or electrons, which is a net charge for an atom or molecule. Neutrons are also composed of atoms but are neutral. An ion is just an atom or a molecule with a charge, and it’ll have a charge if the protons are not equal to electrons. That is because there’s also a relative increase in the effective nuclear charge. Lithium is positive 520 kilojoules per mole, and Beryllium’s goes up to 900 kilojoules per mole, and then again, in general, there’s an increase in ionisation energies going over to neon. Looking at the periodic trend, as the students go from lithium over to neon, across the periodic table, the students can notice that there’s an increase in the ionisation energy. It applies to all the elements on the periodic table and not just the atoms that are gases at room temperature. 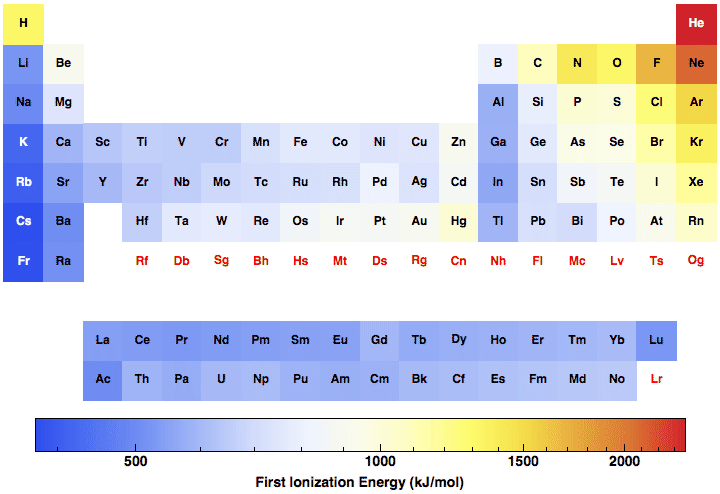 
Ionisation energy is the amount of energy required to remove an electron from a specific gaseous atom or ion.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
